 
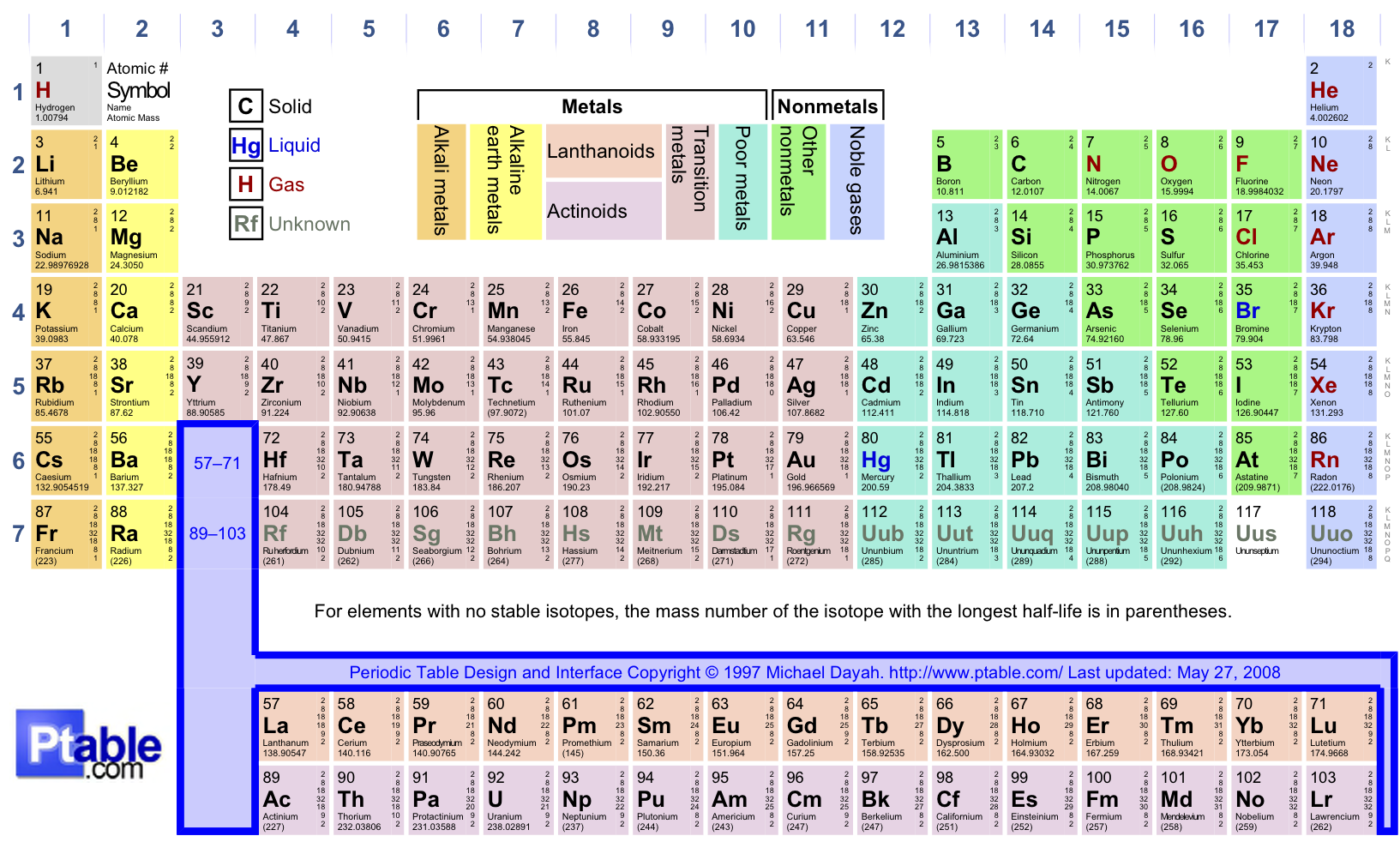
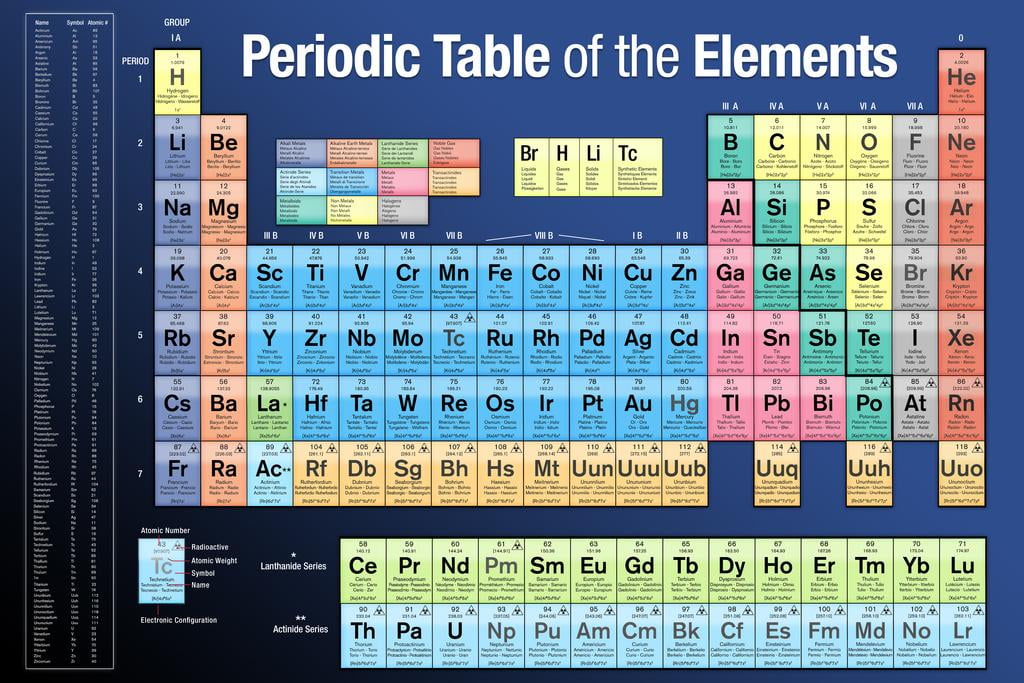
The first two used a combination of Roman numerals and letters. Through the years, three different systems have been used to number the groups on a Periodic Table. Families of elements on the periodic table are in vertical columns called groups. Today's atomic theory also supports orbitals in molecules.Ī timeline of chemical element discoveries. Within an electron energy sublevel on the Periodic Table, orbitals are represented by two element squares. These energy overlaps will be explained in the next concept.Īn atomic orbital can hold one or two electrons.Even though the first elements in the f sublevel on the Periodic Table are on the fifth row, the first electron energy level to have an f sublevel is the fourth energy level.Even though the first elements in the d sublevel on the Periodic Table are on the fourth row, the first electron energy level to have a d sublevel is the third energy level.Both of these sublevels overlap the s sublevel of the next lowest electron energy level. One thing that does not show on the Periodic Table is the overlap of the d and f sublevels. g -block (on the extended periodic table).These "areas" on the Periodic Table are often refered to as "blocks": f sublevel - the fourteen columns of two below the main body of the table.d sublevel - the ten short columns in the middle of the table.p sublevel - the six tall columns on the right, without helium.For the purpose of reading sublevels, it should be thought of as sitting in the second column beside hydrogen. Helium is at the far right because it is inert (like the other Nobel Gases). s sublevel - the two tall columns on the left.The shape of the Periodic Table makes the four sublevels easy to see. The horizontal rows are numbered 1 to 7 at the extreme left of the table. The number of electron energy levels in an atom is indicated by the horizontal row on which the element is found on the Periodic Table. These elements are often called metalloids. In the area of the dividing line, some elements have both metallic and nonmetallic properties. When moving from left to right across the Periodic Table, metal characteristics decrease and nonmetal characteristics increase. Metals are left of the line, nonmetals are to the right. The dividing line begins between boron and aluminum and stair-steps down and to the right, one square at a time. Many, but not all, Periodic Tables have a line dividing these two. The International Union of Pure and Applied Chemistry (IUPAC) now assigns a temporary systematic name and a 3-letter symbol to new or theoretical elements. 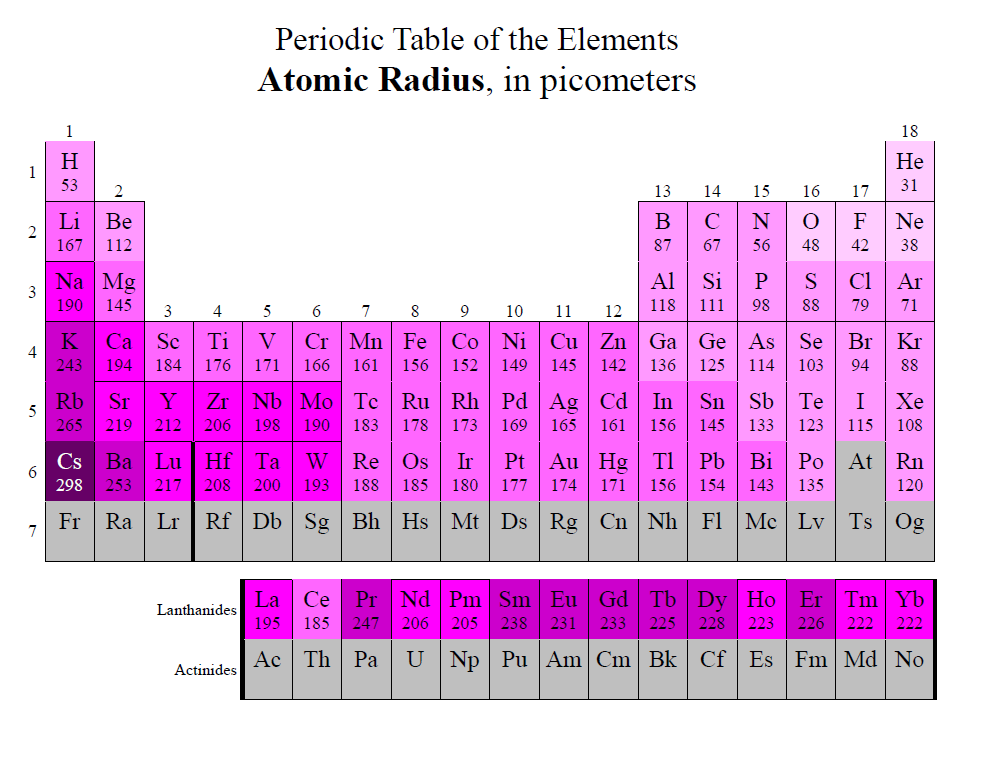
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
